Splenic infarction is a rare clinical condition characterized by ischemic necrosis of splenic tissue, which can lead to potentially life-threatening complications such as rupture, hemorrhage, sepsis, or abscess formation. Infectious causes of splenic infarction are uncommon, and reported viral etiologies include cytomegalovirus, parvovirus B19, adenovirus, and, more recently, COVID-19; however, Epstein-Barr virus (EBV) is rarely associated with this complication.1,2 The pathophysiology of splenic infarction in the setting of EBV infection is not fully understood, but it may involve an intricate interplay of immune responses, transient activation of coagulation pathways, and local vascular dynamics.3 Given its potentially severe consequences, a high index of clinical suspicion is essential for timely diagnosis and management. The aim of this case report is to increase clinicians' awareness about this rare but significant complication of EBV infection.
Case Report
A 28-year-old man with no significant past medical history presented to the emergency department with a five-day history of fever and sore throat. He denied smoking and illicit drug use, and his family history was negative for thromboembolic events. Clinical examination revealed fever (38.5 °C) with sinus tachycardia (112 beats per minute) and bilateral palpable superficial cervical lymph nodes with no signs of palpable spleen or liver.
Baseline laboratory test results are listed at Table 1. Key findings included a reversed neutrophil-to-lymphocyte ratio, elevated liver enzymes, and increased lactate dehydrogenase (LDH). Immunoglobulin M antibodies were positive specific to EBV, indicating an acute infection. The patient was admitted. Polymerase chain reaction (PCR) testing of a blood sample confirmed the diagnosis of an acute EBV infection. Abdominal ultrasound was negative for abnormalities including hepatosplenomegaly.
On the fifth day of hospitalization, the patient complained of new-onset left upper quadrant abdominal pain. Clinical examination revealed mild tenderness in the left upper quadrant and a palpable spleen. Computed tomography (CT) of the abdomen with intravenous contrast demonstrated multiple enlarged iliac lymph nodes, hepatomegaly (178 mm in the midclavicular line), and moderate splenomegaly (140 mm in the longitudinal axis). Multiple hypodense splenic lesions were visible without contrast enhancement, suggestive of splenic infarcts [Figures 1 and 2]. CT angiography of the abdomen showed no thrombi in the splenic vasculature. Hypercoagulability workup revealed mildly reduced protein S activity. Molecular testing for inherited thrombophilia (Factor V Leiden, Factor II-prothrombin, Factor XIII, MTHFR, and JAK2 V617 gene mutations) yielded negative results [Table 1]. Transesophageal echocardiography showed no evidence of thrombi, vegetation, or other abnormalities.
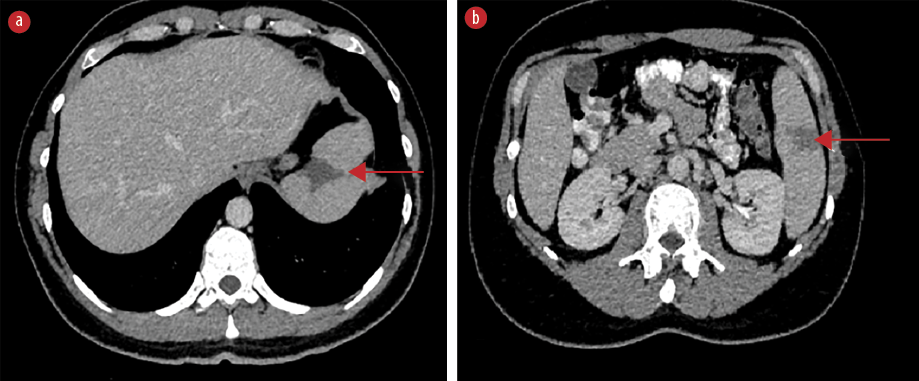 Figure 1: Axial contrast-enhanced CT scan demonstrating wedge-shaped hypodense splenic lesions without contrast enhancement indicative of splenic infarcts (red arrows), (a) located at the T10 level and at (b) the
Figure 1: Axial contrast-enhanced CT scan demonstrating wedge-shaped hypodense splenic lesions without contrast enhancement indicative of splenic infarcts (red arrows), (a) located at the T10 level and at (b) the
L2 level.
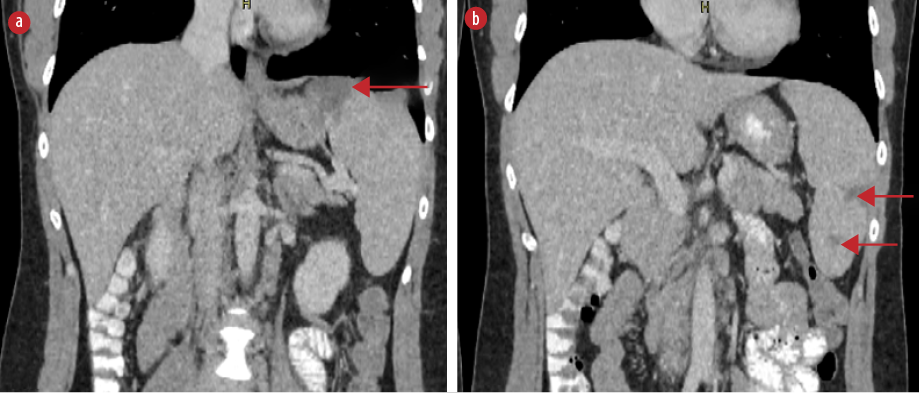 Figure 2: Coronal abdominal CT scan with intravenous contrast demonstrating multiple splenic infarcts (red arrows), (a) located at the superior, as well as (b) the middle and lower parts of the spleen.
Figure 2: Coronal abdominal CT scan with intravenous contrast demonstrating multiple splenic infarcts (red arrows), (a) located at the superior, as well as (b) the middle and lower parts of the spleen.
Table 1: Patient’s laboratory investigation results.
|
White blood cells, K/μL
|
7.8
|
4.7
|
4.1
|
4–10
|
|
Lymphocytes, K/μL
|
4.4
|
2.1
|
2.2
|
1.2–3.8
|
|
Monocytes, K/μL
|
0.3
|
0.4
|
0.4
|
0.2–1
|
|
Platelets, K/μL
|
220
|
265
|
273
|
140–440
|
|
INR
|
1.11
|
1.09
|
1.03
|
0.85–1.15
|
|
Fibrinogen, mg/dL
|
265
|
257
|
278
|
180–400
|
|
aPTT, sec
|
27.9
|
29.2
|
32.4
|
26–36
|
|
EBV IgM
|
5.03
|
-
|
-
|
< 1
|
|
EBV IgG
|
0.01
|
-
|
-
|
< 1
|
|
PCR EBV (serum), copies/mL
|
9.2 × 103
|
-
|
-
|
2.77 × 103
|
|
AST, IU/L
|
99
|
78
|
47
|
5–40
|
|
ALT, IU/L
|
280
|
314
|
104
|
5–45
|
|
LDH, IU/L
|
466
|
303
|
223
|
100–230
|
|
β2 glycoprotein IgM, IgG, U/mL
|
-
|
0.87/3.53
|
-
|
< 20
|
|
Lupus anticoagulant, sec
|
-
|
30.5/–
|
-
|
30.5–40.6
|
|
Anticardiolipin IgG, IgM, U/mL
|
-
|
1.83/4.08
|
-
|
< 7
|
|
Protein C activity, %
|
-
|
82.1%
|
-
|
70–140
|
|
Protein S activity, %
|
-
|
51%
|
-
|
58–127.5
|
|
Factor V Leiden mutation
|
-
|
Negative
|
-
|
-
|
|
Prothrombin gene mutation
|
-
|
Negative
|
-
|
-
|
|
Antithrombin III activity, %
|
-
|
81.3
|
-
|
75–125
|
|
MTHFR gene mutation
|
-
|
Negative
|
-
|
-
|
INR: international normalized ratio; aPTT: activated partial thromboplastin time; EBV: Epstein-Barr virus; IgM: immunoglobulin M; IgG: immunoglobulin G; PCR: polymerase chain reaction; AST: aspartate transaminase; ALT: alanine transaminase; LDH: lactate dehydrogenase.
The patient was managed conservatively with intravenous fluids, analgesics, and prophylactic anticoagulation therapy with apixaban 5 mg daily orally. A significant clinical improvement in the abdominal pain was noted after two days. The patient was discharged after 15 days of hospital stay. He was counseled to avoid strenuous physical activities due to the risk of splenic rupture, and to refrain from hepatotoxic medications for minimum one month. He is under close follow-up and scheduled for an abdominal CT scan to evaluate the evolution of the splenic infarcts.
An informed consent was obtained from the patient.
Discussion
EBV is the most common cause of infectious mononucleosis, affecting up to 95% of the healthy population.4 The main symptoms include fever, fatigue, sore throat, cervical lymphadenopathy, and hepatosplenomegaly.5 Splenic infarction is an extremely rare complication of acute EBV infection, with only 33 cases reported since 1990.6 Most of these cases have involved young adults without an inherited predisposition to a prothrombotic state.3,6
The exact mechanism of splenic infarcts due to EBV remains unknown. Various hypotheses have been proposed, including insufficient blood supply to the enlarged spleen during the acute phase,3,4,7 increased B-cell-mediated immune response,1,4,6 and transient activation of the coagulation cascade.1,4
The clinical presentation of splenic infarction due to EBV typically involves vague and non-specific symptoms, including left upper quadrant pain, splenomegaly, fever, vomiting or chest pain, which may occasionally radiate to the left shoulder.5,6,8 Rarely, it may present as a potentially life-threatening hemorrhage, while some patients remain asymptomatic and are diagnosed incidentally.9 Typically, the manifestation of splenic infarction occurs one to three weeks following the initial onset of infection mononucleosis symptoms.5 Our patient exhibited signs indicative of splenic infarcts ten days after symptom onset.
Laboratory examinations typically reveal mild leukocytosis, elevated LDH, and a transient hypercoagulable state.1,6 Additionally, EBV infection may affect platelet parameters, leading to elevated platelet count or alterations in the mean platelet volume-to-platelet count ratio. Notably, an elevated mean platelet volume-to-platelet count ratio is recognized as a diagnostic marker for liver function damage risk in EBV-infected patients,10 while mean platelet volume is implicated in vascular complications.11 Therefore, EBV should be considered among the primary differential diagnoses when these parameters are affected.12 In our case, these parameters were meticulously monitored and remained within normal limits throughout the patient’s hospital stay.
This hypercoagulable state is additionally indicated by the presence of lupus anticoagulant, anticardiolipin antibodies (ACA), antiphospholipid antibodies, or reduced activity of protein C and protein S.1,6,8 The correlation between the elevation of ACA and the occurrence of splenic infarcts in individuals with EBV infection has been documented in the literature.13 Our patient exhibited an elevated LDH, while the hypercoagulability workup was normal, except for a slightly reduced activity of protein S.
Abdominal ultrasound is not recommended as the sole imaging modality because of its low sensitivity.6,9,14 The imaging modality of choice is contrast-enhanced CT, on which splenic infarcts commonly appear as wedge-shaped hypodense lesions without post-contrast enhancement after contrast administration.6,7 On magnetic resonance imaging, splenic infarcts are visualized as lesions with low signal intensity on both T1-weighted and T2-weighted images.9 In the present case, abnormal lesions suggestive of infarcts became evident on CT scan with intravenous contrast as wedge-shaped lesions without contrast enhancement.
Symptomatic management with intravenous fluids, analgesics, and prophylactic anticoagulation on a case-by-case basis is the treatment of choice.6,15 Close follow-up is recommended for at least four weeks, or until resolution of splenomegaly or infarcts, due to the risk of hemorrhage, splenic rupture, abscess, or pseudocyst formation.9,15,16
To our knowledge, this is the 32nd reported case of splenic infarction associated with an EBV infection since 1990.6,17 Most patients were otherwise healthy adults who were diagnosed while evaluating new-onset abdominal pain.6 A transient hypercoagulable state was observed in most cases, as indicated by various affected parameters, including elevated D-dimers, antiphospholipid antibodies, lupus anticoagulant, ACA, β2-glucoprotein I, and decrease protein C or S activity or antithrombin III, among others.6,15 CT scan was the predominant diagnostic tool in most cases, and the management focused primarily on symptomatic interventions.5,15
Given the widespread prevalence of EBV infection in the general population, and the fact that only a limited subset of patients undergo abdominal imaging procedure during the course of their infection, there is a substantial possibility that cases of splenic infarction secondary to EBV remain undiagnosed, and therefore underreported.7 Continuous clinical vigilance and reporting of such cases are vital to gain a better understanding of the predisposing factors and clinical presentation, and to optimize treatment practices for splenic infarctions associated with EBV infection.
Conclusion
Splenic infarction is an extremely rare complication of EBV infection, with potentially severe complications such as splenic rupture, abscess, or pseudocyst formation. Raised clinical suspicion of this complication is crucial, particularly in patients presenting with abdominal pain and a recent history of EBV infection.
Disclosure
The authors declare no conflicts of interest.
references
- 1. Li Y, George A, Arnaout S, Wang JP, Abraham GM. Splenic infarction: an under-recognized complication of infectious mononucleosis? Open Forum Infect Dis 2018 Feb;5(3):ofy041.
- 2. Childers J, Do TV, Smith F, Vangara A, Ganti SS, Akella R. Incidental and asymptomatic splenic infarction and infrarenal thrombus in a COVID-19 patient. Cureus 2022 Jul 4;14(7):e26555.
- 3. Gavriilaki E, Sabanis N, Paschou E, Grigoriadis S, Mainou M, Gaitanaki A, et al. Splenic infarction as a rare complication of infectious mononucleosis due to Epstein-Barr virus infection in a patient with no significant comorbidity: case report and review of the literature. Scand J Infect Dis 2013 Nov;45(11):888-890.
- 4. Turrian U. Splenic infarction after Epstein-Barr virus infection: case report. Ann Clin Case Rep 2021;6(1):2045.
- 5. Toti JM, Gatti B, Hunjan I, Kottanattu L, Faré PB, Renzi S, et al. Splenic rupture or infarction associated with Epstein-Barr virus infectious mononucleosis: a systematic literature review. Swiss Med Wkly 2023 May;153(5):40081.
- 6. Mamo G, Erickson S, Komanduri K, Zayas D, Aggarwal N. Infectious mononucleosis-induced splenic infarction: perhaps more common in healthy individuals than previously thought. Cureus 2023 May 25;15(5):e39472.
- 7. Antopolsky M, Hiller N, Salameh S, Goldshtein B, Stalnikowicz R. Splenic infarction: 10 years of experience. Am J Emerg Med 2009 Mar;27(3):262-265.
- 8. Machado C, Melo Salgado J, Monjardino L. The unexpected finding of a splenic infarction in a patient with infectious mononucleosis due to Epstein-Barr virus. BMJ Case Rep 2015 Nov;2015:bcr2015212428.
- 9. Naviglio S, Abate MV, Chinello M, Ventura A. Splenic infarction in acute infectious mononucleosis. J Emerg Med 2016 Jan;50(1):e11-e13.
- 10. Han X, Xu P, Duan X, Liu Y, Zhang J, Xu H. High mean platelet volume-to-platelet count ratio as a diagnostic maker for increased risk of liver function damage in pediatric patients with infectious mononucleosis in China. Exp Ther Med 2019 Dec;18(6):4523-4527.
- 11. Abd El-Hameed MR, Abozied AM. Mean platelet volume in impaired fasting glucose subjects and diabetic patients as a risk factor for thrombotic complications. J Am Sci 2013;9(9):12-17.
- 12. Almiş H. Evaluation of platelet parameters in children with primary Epstein-Barr virus infection. Ann Clin Anal Med 2017;8(5).
- 13. Hunt DP, Thabet A, Rosenberg ES. Case 29-2010: a 29-year-old woman with fever and abdominal pain. New England Journal of Medicine 2010 Sep 23;363(13):1266-1274.
- 14. Batista M, Silva C, Gonçalves FM, Machado M, Freitas S, Silva J, et al. Splenic infarction: a rare complication of infectious mononucleosis. Cureus 2023 Apr;15(4):e37414.
- 15. Singhal A, Suchman K, Rhee A, Agrawal V, Cohen J. Splenic infarction due to Epstein-Barr virus infectious mononucleosis: case report and clinical review. Preprints; 2023 Mar [cited 2023 Jul 9]. Available from: https://www.authorea.com/users/599840/articles/631711-splenic-infarction-due-to-epstein-barr-virus-infectious-mononucleosis-case-report-and-clinical-review?commit=d8bcd914a023f4e947b29efe9ae672258d53e13f.
- 16. Dhibar DP, Chhabria BA, Gupta N, Varma SC. Isolated splenic cold abscesses in an immunocompetent individual. Oman Med J 2018 Jul;33(4):352-355.
- 17. Kobayashi K, Kishi Y, Serizawa Y, Kimizuka Y, Ueno H. Splenic infarction associated with Epstein-Barr virus in an adult with an anatomic anomaly: a case report. Cureus 2023 Jun 16;15(6):e40530.